Inside Our Lab: Unraveling Cancer's Hidden Mechanisms
Our lab investigates the critical stress-adaptive mechanisms that enable cancer cells to survive, resist therapy, and progress. Through advanced cellular and molecular approaches, we aim to uncover vulnerabilities in ribosome quality control, mitochondrial metabolism, and stress response systems.
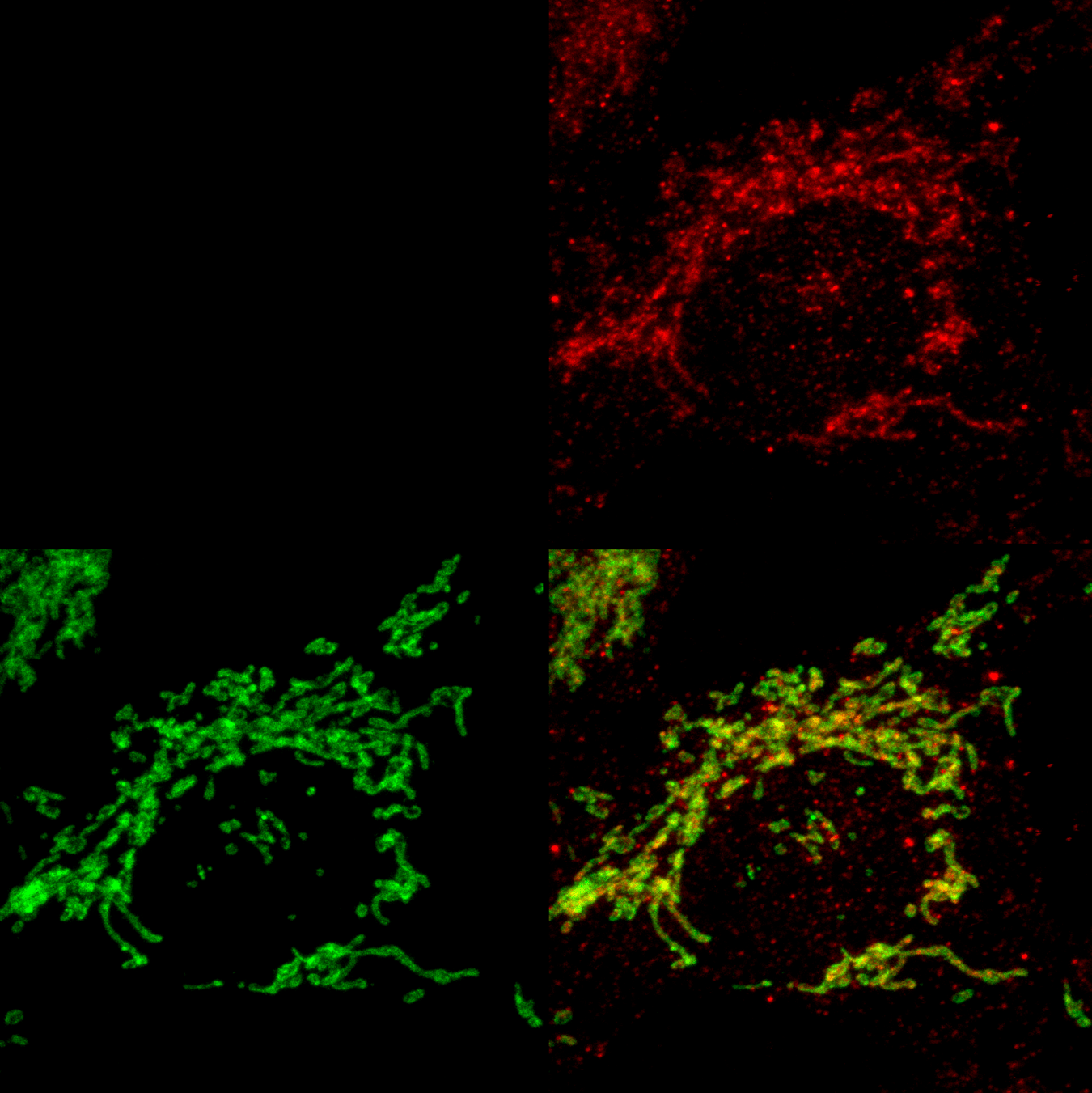
Role of Ribosomal Quality Control in Cancer Progression
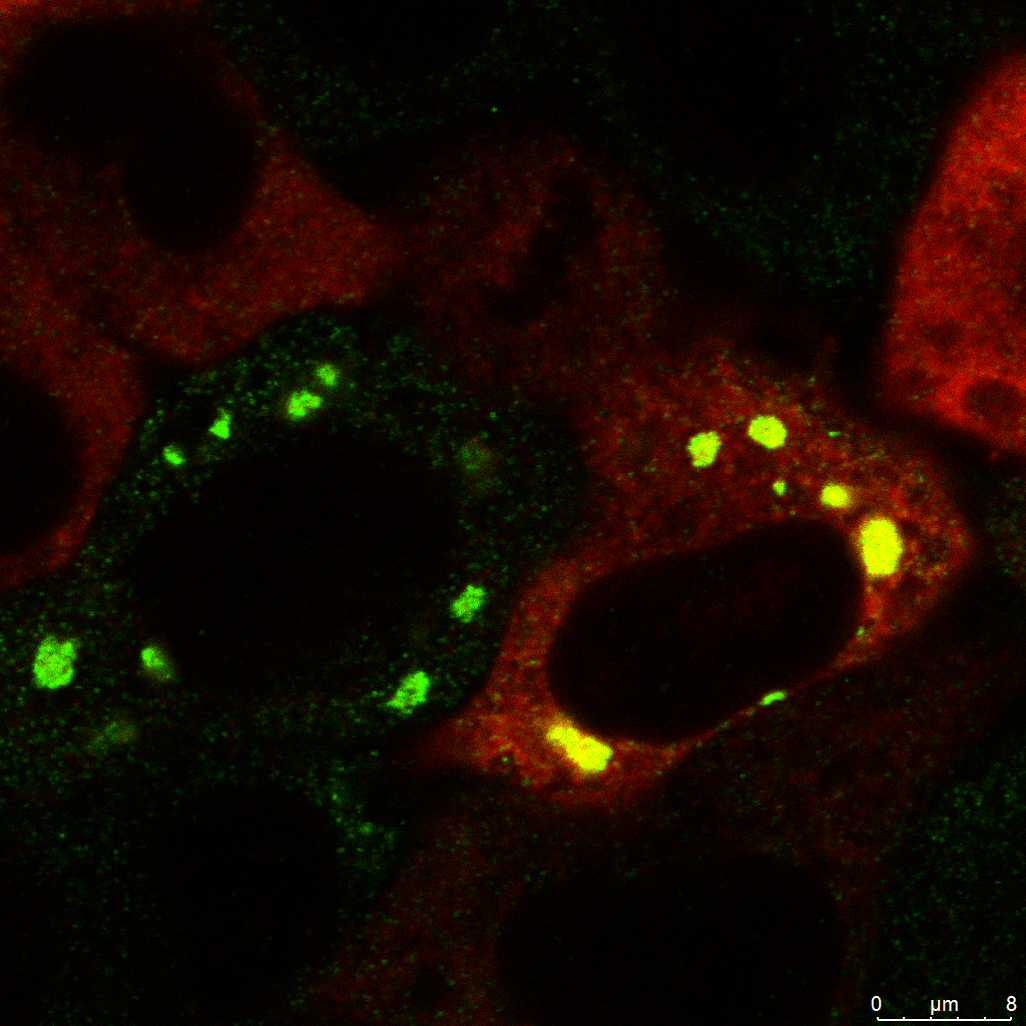
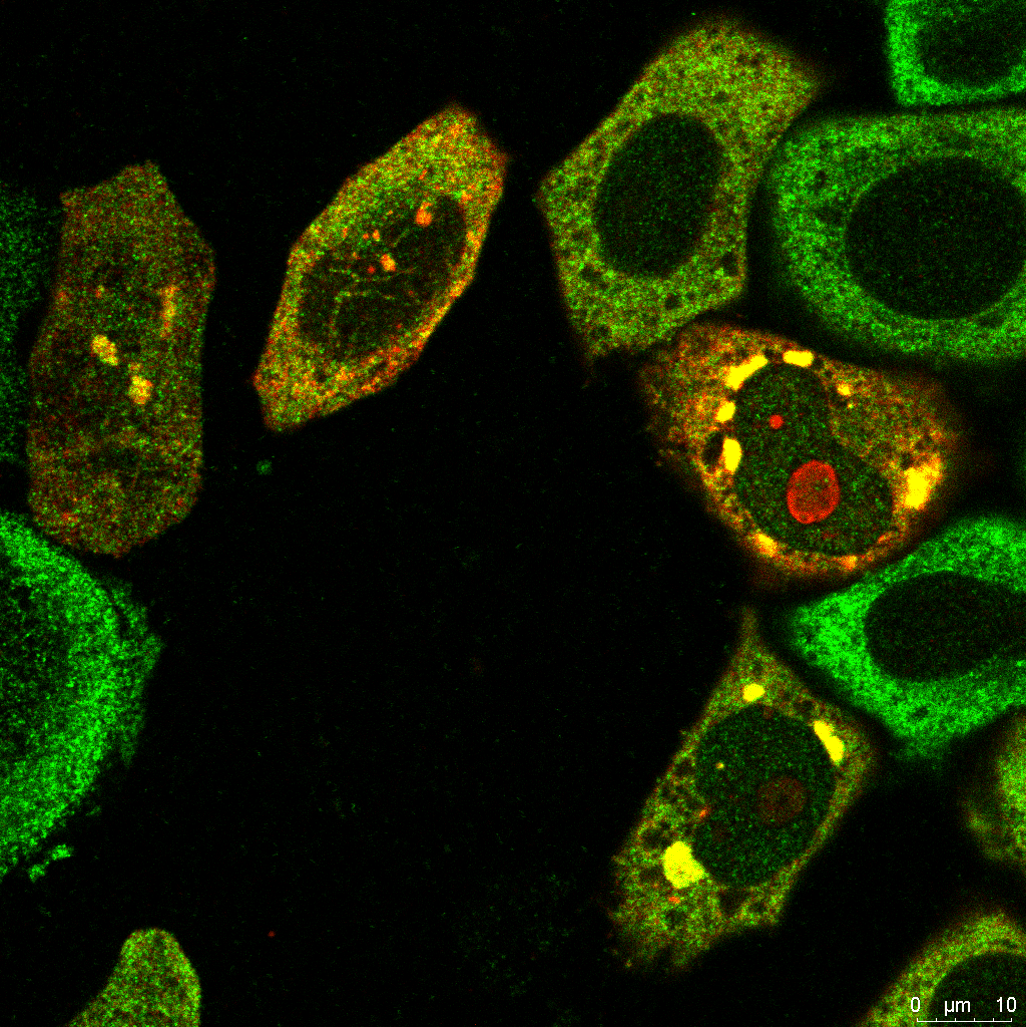
Our research focuses on understanding how the ribosome-associated quality control (RQC) pathway contributes to the development, progression, and therapy resistance of bladder cancer. By using cancer cell lines, cisplatin-resistant models, patient-derived tumor samples, and mice models, we aim to uncover how RQC factors supports cancer cell survival, especially in stem-like and drug-resistant populations. Our goal is to explore the therapeutic potential of targeting RQC mechanisms to overcome chemoresistance and improve treatment strategies in glioblastoma and bladder cancer.
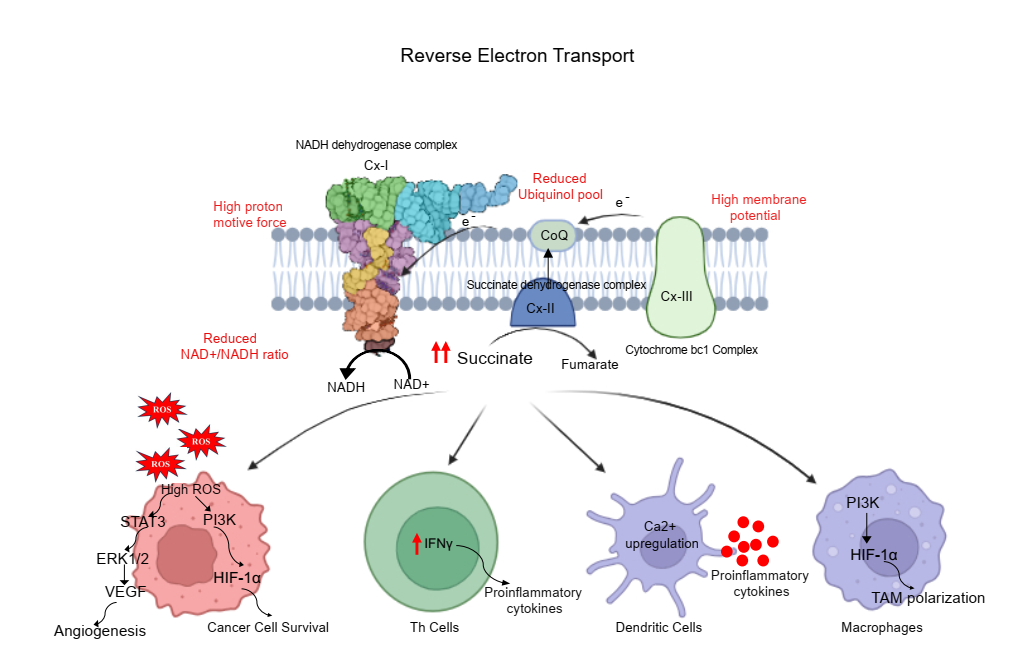
Role of Reverse Electron Transfer in Mediating Metabolic Reprogramming in Cancer
RET is a mitochondrial process where electrons flow backward from ubiquinol to complex I, often driven by a high proton motive force and fueled by metabolic reprogramming in cancer cells. This process leads to elevated reactive oxygen species (ROS) production, which can act as signaling molecules to promote tumor progression, stemness, and therapy resistance. We investigate how RET is regulated in cancer cells, particularly under conditions of nutrient stress, hypoxia, and drug exposure, and how it contributes to metabolic plasticity and redox adaptation. Our studies aim to define RET as a metabolic vulnerability, offering new insights into mitochondrial redox control and its potential as a therapeutic target in aggressive and drug-resistant tumors.
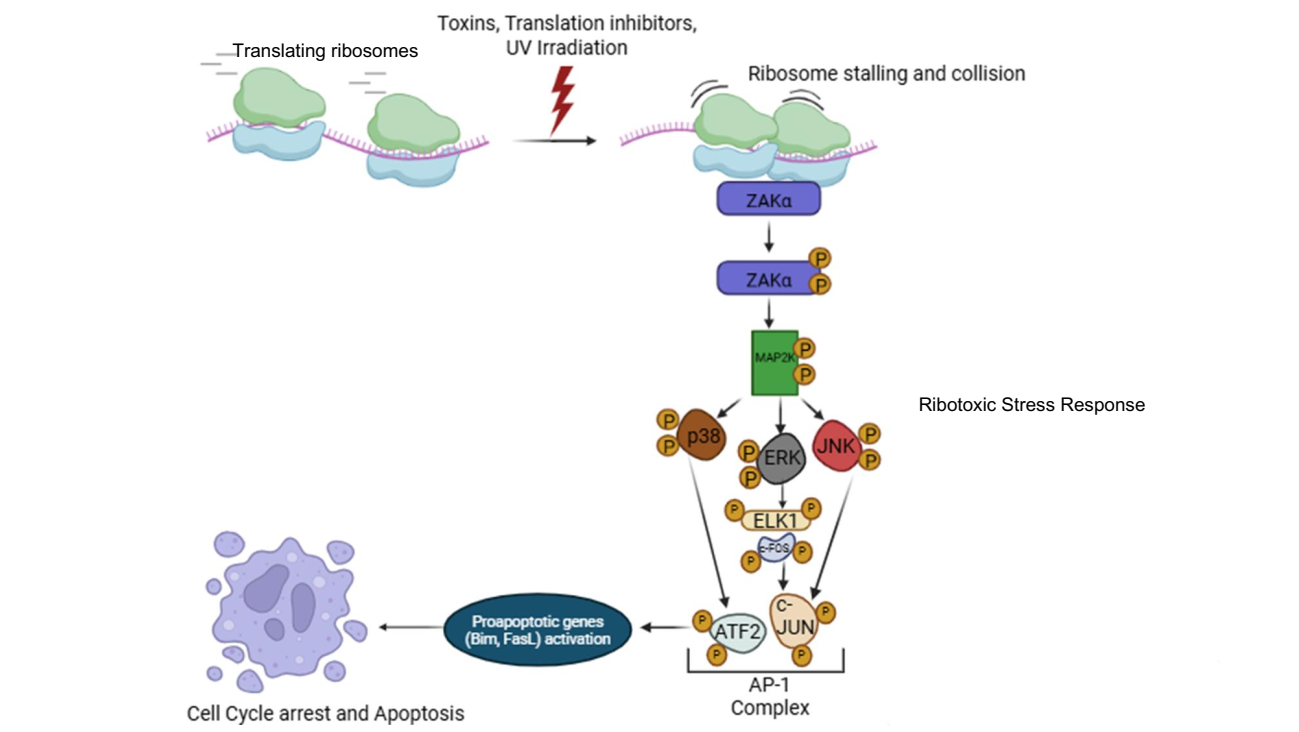
Role of Ribotoxic stress Response Mediated adaptive Mechanism in Cancer Survival during Therapy
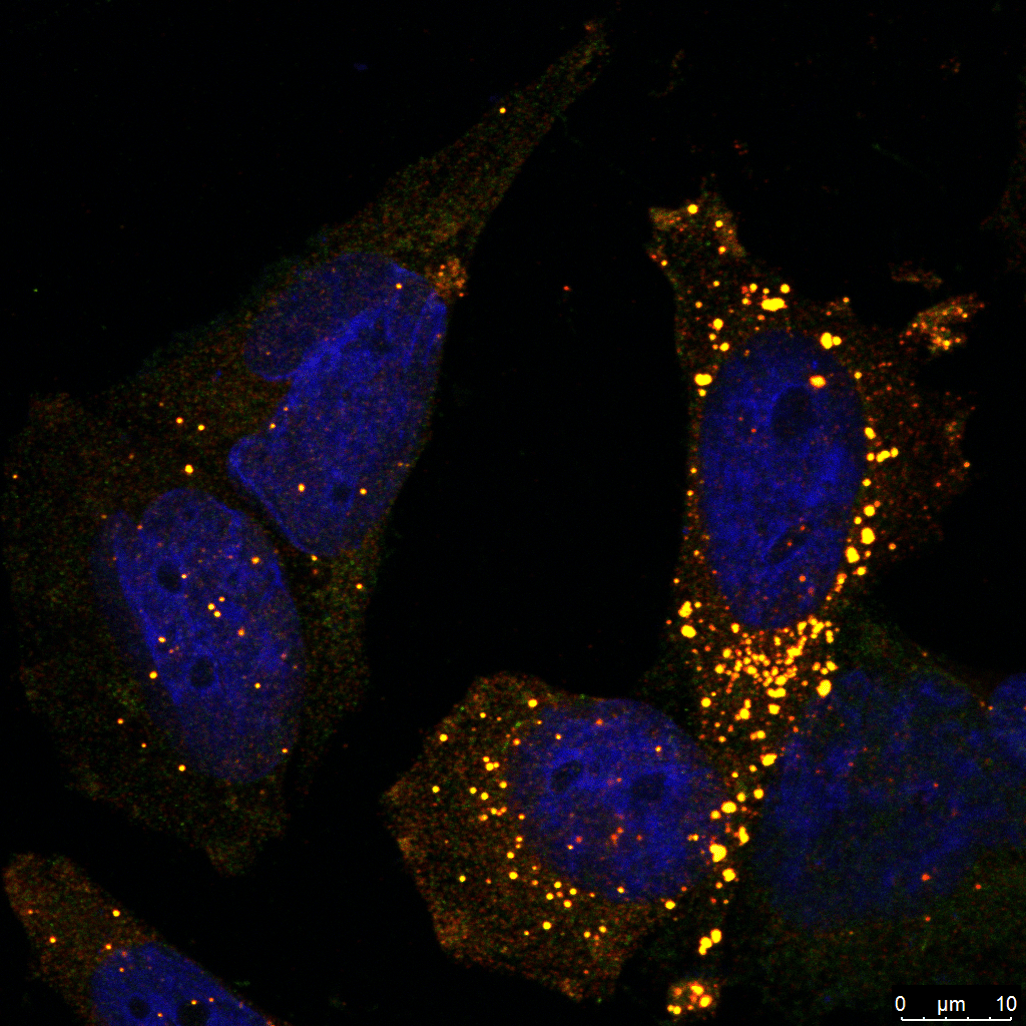
Our research also focuses on ribophagy, a selective form of autophagy that targets damaged or stalled ribosomes for degradation, particularly under conditions of ribotoxic stress. Ribotoxic stress, caused by factors such as oxidative damage, ribosome stalling, or chemical inhibitors, disrupts ribosome function and triggers surveillance pathways like the ribotoxic stress response (RSR) and ribosome-associated quality control (RQC). When these systems are overwhelmed, ribophagy acts as a crucial backup mechanism to maintain ribosome homeostasis and proteostasis. We are investigating how ribophagy is regulated in cancer cells experiencing persistent translation stress, and how it intersects with stress granule dynamics, mTOR signaling, and autophagy pathways. By understanding the role of ribophagy in cancer cell adaptation and survival, our goal is to uncover novel therapeutic opportunities that target ribosome turnover as a strategy to impair tumor resilience and treatment resistance.